Is It Really “Nervous System Medicine”?
Welcome to Ask A Neuroscientist, a new series on Fat Nugs that takes a scientific lens towards some of the biggest and juiciest questions around how canabis work in the body. In the first article of this series, we explore if cannabis actually activates the vagus nerve, if it’s really nervous system medicine, and why it may feel like it – without actually working on the vagus nerve at all.
“CBD has been shown to have a positive effect on the vagus nerve by increasing its activity,” claims one popular CBD wellness brand. Another tincture promises to “activate your parasympathetic nervous system” through “vagal stimulation.”
Scroll through cannabis marketing materials, and you’ll find the vagus nerve invoked everywhere—positioned as the master switch for calm that cannabis can flip on demand.
What is the Vagus Nerve?
The vagus nerve has become a kind of celebrity in modern wellness culture. From breathwork apps to cold plunges to cannabis products marketed as “nervous system medicine,” this cranial nerve is often described as something that can be “activated” like a button.
But anatomically and physiologically, the vagus nerve is not a button. It’s a complex, bidirectional communication highway that plays a far more nuanced role in regulating bodily states than popular discourse suggests.
The vagus nerve (cranial nerve X) is the primary parasympathetic nerve connecting to organs in the chest and abdomen. It originates in the medulla (the lower part of the brainstem) and extends to the heart, lungs, digestive tract, and liver, carrying signals between the brain and internal organs (Breit et al., 2018).
Crucially, approximately 80% of vagal fibers are afferent, meaning they transmit information from the body to the brain, not the other way around (Berthoud & Neuhuber, 2000). This alone complicates the idea that the vagus nerve is primarily a calming “output” pathway that can be directly stimulated to induce relaxation.
Vagal signaling plays a central role in maintaining homeostasis (physiological balance), not tranquility per se. Through its parasympathetic efferent fibers (output pathways), the vagus nerve helps slow heart rate, regulate digestion, and modulate inflammatory responses.
At the same time, vagal afferents (sensory input pathways) continuously relay information about what’s happening in the body—gut activity, blood pressure changes, immune signals. This information flows to brain regions that process body awareness, emotion, and threat response, including the insula and amygdala (Critchley & Harrison, 2013; Breit et al., 2018).
In other words, the vagus nerve is deeply involved in how the body informs the brain about safety and danger, not simply how the brain enforces calm.
Understanding Vagal Tone
The term “vagal tone” is often used as shorthand for nervous system health, but its scientific meaning is more specific and limited. Vagal tone refers to the influence of the parasympathetic nervous system—via the vagus nerve—on heart rate variability (HRV), particularly respiratory sinus arrhythmia (the natural variation in heart rate with breathing) (Porges, 2007).
Higher resting vagal tone is generally associated with greater autonomic flexibility (the ability to shift smoothly between arousal and calm), emotional regulation, and resilience to stress (Thayer & Lane, 2000). Importantly, this does not mean that “higher vagal tone” equals constant relaxation. Rather, it reflects the nervous system’s capacity to respond efficiently to changing demands.
Heart rate variability is the most commonly used proxy for vagal tone, but it is an imperfect measure. HRV reflects the interplay of multiple physiological systems, including respiration, blood pressure regulation, and sympathetic activity, not just vagal output (Shaffer & Ginsberg, 2017).
While vagal withdrawal and re-engagement shape HRV patterns, HRV alone cannot definitively indicate vagus nerve activation or parasympathetic dominance in a given moment. This matters when evaluating claims that a substance or practice “stimulates the vagus nerve” based solely on subjective calm or indirect physiological markers.
The popular association between the vagus nerve and emotional safety has been further shaped by Polyvagal Theory, which proposes that the vagus nerve supports social engagement and defensive states through distinct neural circuits (Porges, 2011).
The Contraversial Polyvagal Theory
While influential in trauma therapy communities, Polyvagal Theory remains controversial, particularly regarding its anatomical claims and evolutionary interpretations (Grossman & Taylor, 2007). Nonetheless, even critics agree on a central point: vagal signaling is context-sensitive and state-dependent. It does not operate as a simple on/off switch for calm.
Here’s what the anatomy and physiology of the vagus nerve actually show: the vagus nerve is not a direct pathway to relaxation, nor is vagal tone synonymous with feeling good. It’s a dynamic communication system that integrates bodily signals, emotional appraisal, and autonomic regulation.
Any claim that cannabis—or any intervention—acts as “vagus nerve medicine” must be evaluated against this biological reality. Before asking what cannabis does to the vagus nerve, we first have to understand what the vagus nerve actually does.
The Endocannabinoid System & Autonomic Regulation
If cannabis does not directly stimulate the vagus nerve, why does it so often feel like a nervous system regulator? The answer lies not in cranial nerve anatomy, but in the endocannabinoid system (ECS)—a distributed neuromodulatory network that interfaces with stress physiology, affective processing, and autonomic output across multiple brain and body systems.
The ECS is composed of endogenous ligands (the body’s own signaling molecules, primarily anandamide and 2-arachidonoylglycerol), cannabinoid receptors (CB1 and CB2), and enzymes responsible for ligand synthesis and degradation (Lu & Mackie, 2016).
How and Where the Endocannabinoid System Works
Unlike classical neurotransmitter systems, the ECS operates largely through retrograde signaling (backward communication from the receiving neuron to the sending neuron), allowing postsynaptic neurons to regulate presynaptic activity and maintain synaptic balance (Katona & Freund, 2012). This balancing act is especially relevant in circuits governing stress and autonomic regulation.
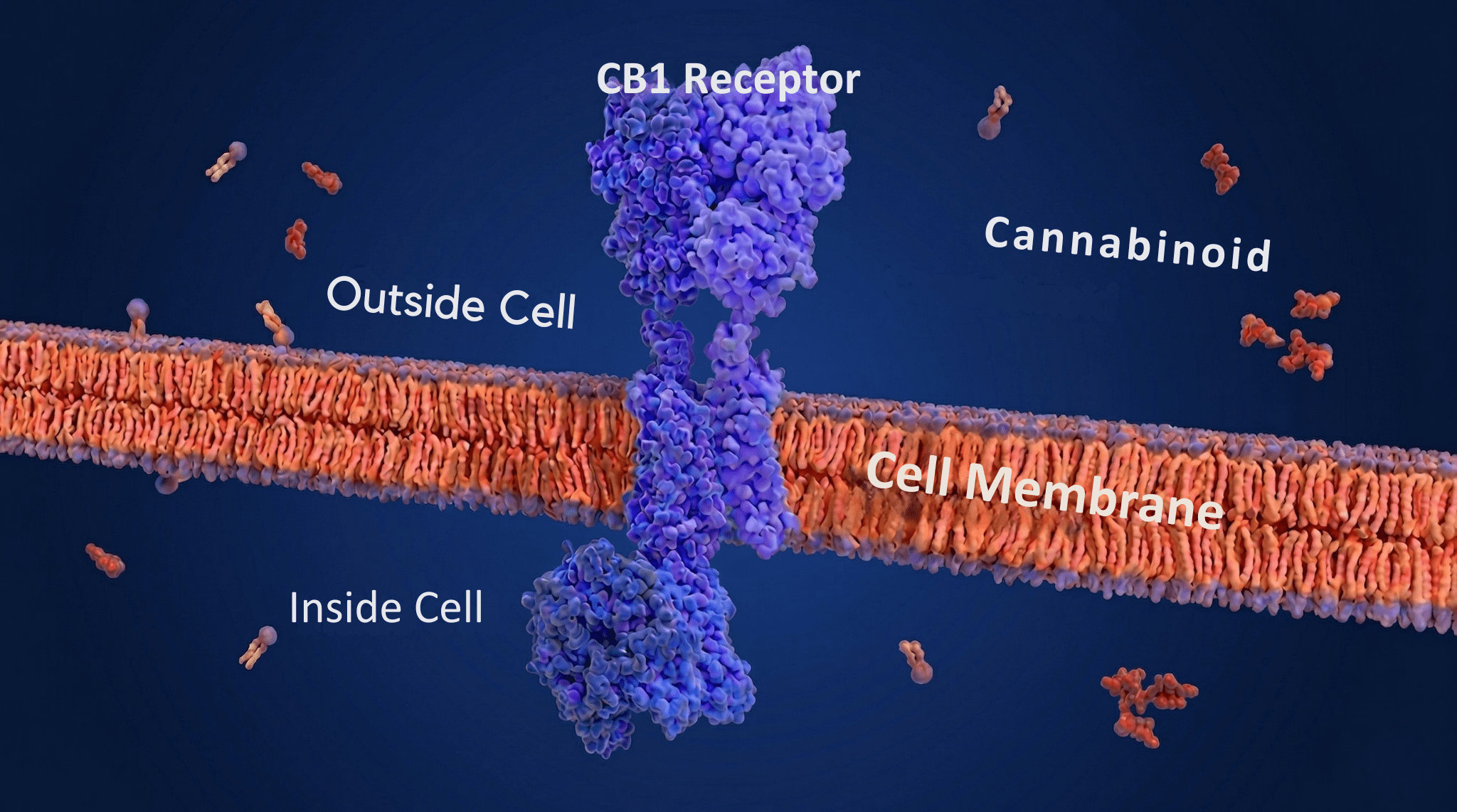
CB1 receptors are densely concentrated throughout the brain, especially in regions that coordinate stress responses: the hypothalamus, amygdala, hippocampus, and brainstem areas controlling heart rate and breathing (Herkenham et al., 1990; Kano et al., 2009).
Through these sites, cannabinoids influence the hypothalamic–pituitary–adrenal (HPA) axis (the body’s main stress response system), emotional threat processing, and downstream sympathetic–parasympathetic balance. Importantly, this modulation occurs upstream of the vagus nerve, shaping how autonomic output is generated rather than directly activating parasympathetic fibers.
The Role of Cannabinoids in the Endocannabinoid System
THC
Δ9-tetrahydrocannabinol (THC), the primary psychoactive component of cannabis, exhibits biphasic effects (opposite effects at low versus high doses) on autonomic function. At low doses, THC has been shown to reduce stress reactivity and attenuate HPA axis activation, whereas higher doses can increase anxiety, sympathetic arousal, and cardiovascular stimulation (Moreira & Wotjak, 2010; Rey et al., 2012).
This dose-dependent reversal is a hallmark of cannabinoid pharmacology and helps explain why cannabis can feel calming in some contexts and destabilizing in others. The same compound can simultaneously dampen perceived threat while elevating heart rate—an autonomic paradox often misattributed to vagal stimulation.
CBD
Cannabidiol (CBD), by contrast, does not bind CB1 receptors with high affinity and instead exerts indirect effects through multiple targets, including serotonin 5-HT1A receptors, TRPV1 channels, and modulation of endocannabinoid degradation (Campos et al., 2012; Blessing et al., 2015).
Preclinical and human studies suggest that CBD may reduce stress-induced autonomic arousal and anxiety without producing the stimulant-like effects associated with THC (Zuardi et al., 2017). These effects are better characterized as stress-buffering than parasympathetic activation, reinforcing the distinction between autonomic regulation and vagal tone per se.
The ECS
The ECS also interacts with immune and inflammatory signaling, which indirectly shapes autonomic balance. CB2 receptors, expressed primarily on immune cells, influence the release of inflammatory signaling molecules (Turcotte et al., 2016). Because inflammation feeds back into autonomic and affective circuits via vagal afferents and central immune signaling, cannabinoid-mediated immunomodulation may alter how the nervous system interprets bodily states of distress or safety (Breit et al., 2018). Again, this represents contextual modulation, not direct vagus nerve stimulation.
Crucially, ECS signaling is state-dependent. Endocannabinoid release increases during stress as a compensatory mechanism to restore equilibrium (Hill et al., 2010). Exogenous cannabinoids can either support or disrupt this process depending on timing, dose, individual neurobiology, and prior stress exposure. Chronic stress, trauma history, and sex hormones have all been shown to influence ECS tone and receptor expression, contributing to the highly individualized autonomic effects of cannabis (Hill et al., 2016).
The ECS functions as a meta-regulatory system, shaping how autonomic responses are generated, interpreted, and resolved. Cannabis does not act on the vagus nerve directly, but it can alter the neural and physiological conditions under which vagal signaling operates.
When people describe cannabis as “nervous system medicine,” they’re often responding to this broader recalibration of stress appraisal and bodily awareness—not to increased parasympathetic output in a narrow anatomical sense. Understanding this distinction is essential for evaluating both the promise and the limits of cannabis as a tool for autonomic regulation.
Why Cannabis Feels Like Vagus Nerve Activation
If cannabis does not directly stimulate the vagus nerve, why do so many people describe its effects using the language of parasympathetic calm: “grounded,” “regulated,” “safe,” or “finally able to breathe”?
The answer lies in how cannabis alters interoception (awareness of internal body states), emotional significance, and the brain’s interpretation of bodily signals, rather than in direct changes to vagal output itself.
Interoception
Interoception—the sensing of internal bodily states—is mediated by brain regions that receive dense input from vagal afferents, particularly the insular cortex and anterior cingulate (Craig, 2002; Critchley & Harrison, 2013).
Cannabis users frequently report increased sensitivity to internal sensations—heartbeat, breath, muscle tension, gut feelings (Seth & Tsakiris, 2018). When these sensations shift toward perceived ease or coherence, the experience can be interpreted subjectively as “vagus nerve activation,” even if autonomic balance has not measurably changed.
Cannabis also alters time perception and attentional focus, both of which influence autonomic experience. THC reliably slows subjective time and narrows mental focus, drawing awareness into present-moment bodily sensations (Sewell et al., 2013). This inward attentional shift resembles the phenomenology of practices like meditation and slow breathing, which are known to increase parasympathetic influence.
However, resemblance does not imply equivalence. Feeling slower, quieter, or more embodied does not necessarily mean the parasympathetic nervous system is dominant; it means the brain is interpreting bodily signals differently.
Threat Appraisal
Another key factor is threat appraisal. Autonomic states are shaped not only by physiological inputs but by cognitive and emotional interpretations of safety and danger (Thayer & Lane, 2000). Cannabis, particularly at lower doses, can reduce perceived threat and dampen amygdala reactivity to stressors (Phan et al., 2008).
When the brain appraises the environment as safer, sympathetic drive may decrease secondarily, producing sensations associated with parasympathetic states. This top-down modulation can feel indistinguishable from direct vagal engagement, even though the mechanism is mental and emotional rather than purely physical.
Trauma History
Trauma history further complicates this picture. Individuals with chronic stress or trauma exposure often exhibit altered autonomic baselines and heightened interoceptive vigilance (van der Kolk, 2014). For these individuals, cannabis-induced changes in bodily perception—such as softened muscle tone, altered breathing rhythms, or emotional numbing—may be experienced as profound relief.
Importantly, relief does not always correspond to increased autonomic flexibility. In some cases, cannabis may reduce distress by blunting sensory or emotional input, rather than by restoring balanced vagal–sympathetic interplay (Bonn-Miller et al., 2011).
Breathwork
Breathing patterns offer another clue. Many users report that cannabis “slows their breathing,” a sensation closely associated with parasympathetic activity. While some studies suggest cannabinoids can influence respiratory rhythm indirectly, these effects are inconsistent and dose-dependent (Pertwee, 2010). More often, the sensation of slower breathing reflects heightened awareness of respiration, not a measurable change in respiratory rate. Increased awareness alone can generate feelings of calm typically attributed to vagal activation.
Finally, cultural narratives shape bodily interpretation. In a wellness landscape saturated with vagus nerve discourse, people are primed to label any state of calm or embodiment as “parasympathetic.” Once a sensation is named, it becomes reinforced. This does not make the experience invalid—but it does blur the line between felt sense and physiological mechanism.
Cannabis feels like vagus nerve activation because it modifies how the brain attends to, interprets, and assigns meaning to bodily signals. It can soften threat perception, enhance interoceptive awareness, and shift attentional focus inward—all experiences commonly associated with parasympathetic states. But these effects arise from central modulation of perception and appraisal, not direct stimulation of the vagus nerve itself. Understanding this distinction allows for more precise language—and safer, more informed use—without dismissing the real relief many people experience.
Clinical & Cultural Implications of Cannabis as Vagus Nerve Medicine
The widespread framing of cannabis as “vagus nerve medicine” may seem benign, even helpful, but imprecise language carries real clinical and cultural consequences.
When complex neurophysiological processes are reduced to a single anatomical shortcut, the result is not just misunderstanding—it can shape expectations, treatment decisions, and patterns of use in ways that undermine nervous system health rather than support it.
Subjective Calm, Not Parasympathetic Dominance
Clinically, conflating subjective calm with parasympathetic dominance risks overselling cannabis as a universal regulator. Autonomic regulation depends on flexibility—the ability to move fluidly between states—not on maintaining constant calm (Thayer & Lane, 2000).
Framing cannabis as a tool that “activates the vagus nerve” may encourage individuals to pursue stillness at the expense of responsiveness, particularly in populations already prone to dissociation or emotional blunting. For trauma-exposed individuals, relief that comes from dampening sensation rather than restoring adaptive engagement can be misinterpreted as healing (van der Kolk, 2014).
From a medical perspective, accuracy matters for dose, indication, and risk communication. THC’s biphasic effects mean that increasing dose does not reliably increase therapeutic benefit and may instead exacerbate anxiety, tachycardia, or sympathetic arousal (Moreira & Wotjak, 2010). When cannabis is marketed as parasympathetic support, patients may overlook these dose-dependent risks or misattribute adverse effects to personal failure rather than pharmacology. Clear language—describing cannabis as a context-dependent stress modulator, not a vagal stimulant—supports safer use and more realistic expectations.
Culturally, vagus nerve discourse reflects a broader trend in wellness spaces: the desire for scientific credibility without mechanistic rigor. Invoking the vagus nerve lends scientific authority to interventions that feel calming, but this shortcut can obscure more meaningful questions about environment, trauma, social safety, and structural stressors.
Nervous systems do not regulate in isolation. Housing insecurity, racialized stress, caregiving burden, and chronic illness all shape autonomic baselines in ways no supplement or plant can override (McEwen & Akil, 2020). Overemphasizing individual “nervous system hacks” risks depoliticizing stress and medicalizing adaptation to unjust conditions.
At the same time, rejecting the vagus nerve narrative entirely would miss an opportunity. The popularity of this language signals a growing public intuition that bodily regulation matters—that mental health is inseparable from physiology.
Cannabis as a Tool, Not a Magic Wand
Rather than dismissing this intuition, clinicians, educators, and writers can refine it. Cannabis can be discussed as a tool that alters interoceptive awareness, threat appraisal, and stress responsiveness, while acknowledging that these effects vary widely across individuals and contexts.
Precision is not pedantry; it is harm reduction. Saying that cannabis may support nervous system regulation indirectly—by shifting perception, emotion, and stress signaling—respects both the science and the lived experience of users. It allows room for benefit without promising anatomical outcomes that cannot be reliably delivered. Most importantly, it reframes regulation not as a state to be achieved, but as a capacity to be cultivated over time, with or without cannabis.
In this light, the question is no longer whether cannabis is “vagus nerve medicine,” but whether we are willing to talk honestly about what nervous system support actually requires. The answer lies not in naming a nerve, but in understanding the systems—and the lives—in which that nerve is embedded.
About the Author
RN Collins is a 1L at Northeastern University School of Law and a neuroscientist exploring how brain health and the environment intersect. Through her writing, she bridges academic research and science communication to reframe how psychoactive plants and other traditional and alternative medicines are understood. She’s building a career that connects law, technology, and creativity—and welcomes conversations and opportunities across fields that share that vision. Connect with her on LinkedIn!
References
- Berthoud, H.-R., & Neuhuber, W. L. (2000). Functional and chemical anatomy of the afferent vagal system. Autonomic Neuroscience, 85(1–3), 1–17. https://doi.org/10.1016/S1566-0702(00)00215-0
- Breit, S., Kupferberg, A., Rogler, G., & Hasler, G. (2018). Vagus nerve as modulator of the brain–gut axis in psychiatric and inflammatory disorders. Frontiers in Psychiatry, 9, 44. https://doi.org/10.3389/fpsyt.2018.00044
- Critchley, H. D., & Harrison, N. A. (2013). Visceral influences on brain and behavior. Neuron, 77(4), 624–638. https://doi.org/10.1016/j.neuron.2013.02.008
- Grossman, P., & Taylor, E. W. (2007). Toward understanding respiratory sinus arrhythmia: Relations to cardiac vagal tone, evolution and biobehavioral functions. Biological Psychology, 74(2), 263–285. https://doi.org/10.1016/j.biopsycho.2005.11.014
- Porges, S. W. (2007). The polyvagal perspective. Biological Psychology, 74(2), 116–143. https://doi.org/10.1016/j.biopsycho.2006.06.009
- Porges, S. W. (2011). The polyvagal theory: Neurophysiological foundations of emotions, attachment, communication, and self-regulation. W. W. Norton.
- Shaffer, F., & Ginsberg, J. P. (2017). An overview of heart rate variability metrics and norms. Frontiers in Public Health, 5, 258. https://doi.org/10.3389/fpubh.2017.00258
- Thayer, J. F., & Lane, R. D. (2000). A model of neurovisceral integration in emotion regulation and dysregulation. Journal of Affective Disorders, 61(3), 201–216. https://doi.org/10.1016/S0165-0327(00)00338-4
- Blessing, E. M., Steenkamp, M. M., Manzanares, J., & Marmar, C. R. (2015). Cannabidiol as a potential treatment for anxiety disorders. Neurotherapeutics, 12(4), 825–836. https://doi.org/10.1007/s13311-015-0387-1
- Campos, A. C., Moreira, F. A., Gomes, F. V., Del Bel, E. A., & Guimarães, F. S. (2012). Multiple mechanisms involved in the large-spectrum therapeutic potential of cannabidiol in psychiatric disorders. Philosophical Transactions of the Royal Society B, 367(1607), 3364–3378. https://doi.org/10.1098/rstb.2011.0389
- Herkenham, M., Lynn, A. B., Johnson, M. R., Melvin, L. S., de Costa, B. R., & Rice, K. C. (1990). Characterization and localization of cannabinoid receptors in rat brain: A quantitative in vitro autoradiographic study. Journal of Neuroscience, 10(6), 1900–1916. https://doi.org/10.1523/JNEUROSCI.10-06-01900.1990
- Hill, M. N., McLaughlin, R. J., Bingham, B., Shrestha, L., Lee, T. T. Y., Gray, J. M., … Gorzalka, B. B. (2010). Endogenous cannabinoid signaling is essential for stress adaptation. Proceedings of the National Academy of Sciences, 107(20), 9406–9411. https://doi.org/10.1073/pnas.0914661107
- Hill, M. N., Patel, S., Carrier, E. J., Rademacher, D. J., Ormerod, B. K., Hillard, C. J., & Gorzalka, B. B. (2016). Downregulation of endocannabinoid signaling in the hippocampus following chronic unpredictable stress. Neuropsychopharmacology, 41(1), 215–227. https://doi.org/10.1038/npp.2015.156
- Kano, M., Ohno-Shosaku, T., Hashimotodani, Y., Uchigashima, M., & Watanabe, M. (2009). Endocannabinoid-mediated control of synaptic transmission. Physiological Reviews, 89(1), 309–380. https://doi.org/10.1152/physrev.00019.2008
- Katona, I., & Freund, T. F. (2012). Multiple functions of endocannabinoid signaling in the brain. Annual Review of Neuroscience, 35, 529–558. https://doi.org/10.1146/annurev-neuro-062111-150420
- Lu, H.-C., & Mackie, K. (2016). An introduction to the endogenous cannabinoid system. Biological Psychiatry, 79(7), 516–525. https://doi.org/10.1016/j.biopsych.2015.07.028
- Moreira, F. A., & Wotjak, C. T. (2010). Cannabinoids and anxiety. Current Topics in Behavioral Neurosciences, 2, 429–450. https://doi.org/10.1007/7854_2009_6
- Rey, A. A., Purrio, M., Viveros, M. P., & Lutz, B. (2012). Biphasic effects of cannabinoids in anxiety responses: CB1 and GABA(B) receptors in the balance of GABAergic and glutamatergic neurotransmission. Neuropsychopharmacology, 37(12), 2624–2634. https://doi.org/10.1038/npp.2012.123
- Turcotte, C., Blanchet, M.-R., Laviolette, M., & Flamand, N. (2016). The CB2 receptor and its role as a regulator of inflammation. Cellular and Molecular Life Sciences, 73(23), 4449–4470. https://doi.org/10.1007/s00018-016-2300-4
- Zuardi, A. W., Rodrigues, N. P., Silva, A. L., Bernardo, S. A., Hallak, J. E. C., Guimarães, F. S., & Crippa, J. A. S. (2017). Inverted U-shaped dose–response curve of the anxiolytic effect of cannabidiol during public speaking in real life. Frontiers in Pharmacology, 8, 259. https://doi.org/10.3389/fphar.2017.00259
- Bonn-Miller, M. O., Vujanovic, A. A., & Zvolensky, M. J. (2011). Emotional dysregulation: Association with coping-oriented marijuana use motives among current marijuana users. Substance Use & Misuse, 46(6), 685–692. https://doi.org/10.3109/10826084.2010.537733
- Craig, A. D. (2002). How do you feel? Interoception: The sense of the physiological condition of the body. Nature Reviews Neuroscience, 3(8), 655–666. https://doi.org/10.1038/nrn894
- Pertwee, R. G. (2010). Receptors and channels targeted by phytocannabinoids other than Δ9-THC. British Journal of Pharmacology, 160(3), 523–539. https://doi.org/10.1111/j.1476-5381.2010.00737.x
- Phan, K. L., Angstadt, M., Golden, J., Onyewuenyi, I., Popovska, A., & de Wit, H. (2008). Cannabinoid modulation of amygdala reactivity to social signals of threat in humans. Journal of Neuroscience, 28(10), 2313–2319. https://doi.org/10.1523/JNEUROSCI.5603-07.2008
- Seth, A. K., & Tsakiris, M. (2018). Being a beast machine: The somatic basis of selfhood. Trends in Cognitive Sciences, 22(11), 969–981. https://doi.org/10.1016/j.tics.2018.08.008
- Sewell, R. A., Schnakenberg, A., Elander, J., Radhakrishnan, R., Williams, A., Skosnik, P. D., Pittman, B., Ranganathan, M., & D’Souza, D. C. (2013). Acute effects of THC on time perception in frequent and infrequent cannabis users. Psychopharmacology, 226(2), 401–413. https://doi.org/10.1007/s00213-012-2915-6
- van der Kolk, B. (2014). The body keeps the score: Brain, mind, and body in the healing of trauma. Viking.
- McEwen, B. S., & Akil, H. (2020). Revisiting the stress concept: Implications for affective disorders. Journal of Neuroscience, 40(1), 12–21. https://doi.org/10.1523/JNEUROSCI.0733-19.2019